
Article added / Artikel hinzugefügt 01.10.2021
Generally Articles and Discussions about Osteosarcoma in Dogs
→ Evaluations of phylogenetic proximity in a group of 67 dogs with
osteosarcoma: a pilot study
Article added / Artikel hinzugefügt 01.10.2021
Generally Articles and Discussions about Osteosarcoma in Dogs
→ Canine Periosteal Osteosarcoma
Images added / Abbildungen hinzugefügt 02.05.2019
Generally Sonography Atlas of Dogs →
Cardiovascular system → Pulmonary vessels
New subcategory added / Neue Unterkategorie hinzugefügt 02.05.2019
Generally Sonography Atlas of Dogs →
Cardiovascular system → Pulmonary vessels
Images added / Abbildungen hinzugefügt 01.05.2019
Generally Sonography Atlas of Dogs →
Cardiovascular system → Heart valvular diseases



Axial Osteosarcoma in Dog – Case Report

Castro J. L. C., Santalucia S., Nazareth W., Castro V. S. P., Pires M. V. M., Leme Jr P. T. O., Paula L. R., Ururahy K. C. B., Corrêa L. F. D. and Raiser A. G..
C. CJL, S. S, W. N, P. CVS, M. PMV, O. LJPT, R. PL, B. UKC, . Axial Osteosarcoma in Dog – Case Report. www.scopemed.org/?mno=32272 [Access: October 08, 2015].

Abstract
Osteosarcoma is the most frequent bone tumor in dogs representing about 85% of the tumors in bones. It’s
occurrence in the axial skeleton is rare and it’s more frequent in females. The prognosis is poor with a great
metastatic occurrence. A white shepherd dog, male, 8 years with a large tumor in the frontal
region, quick growth, rigid consistency and pain by manipulation, was brought to the clinic. Surgical excision was performed and histopathological examination confirmed the suspected
diagnosis as osteosarcoma. The patient was referred for chemotherapy. The tumor showed up again in the same location requiring new surgical procedure, but there was never evidence of metastasis
in this case.
Introduction
Osteosarcoma (OSA) or osteogenic sarcoma (Daleck et al., 2006) is the most common primary bone cancer
diagnosed in dogs, a malignant mesenchymal tumor from the primitive anaplastic bone cell,
characterized by an extracellular matrix osteoid production, which is their histological differentiation
(Neuwald et al., 2006). It represents around 85% of the bone tumors (Costa et al., 2001, Gomes et al., 2008, Oliveira and Silveira, 2008). Its incidence in the axial
skeleton is 25% (Cavalcanti et al., 2004), most reported in females (Daleck et al.,
2006) and occurs principally in older dogs of medium size (Neuwald et al., 2006).
In a study of 116 dogs affected with axial osteosarcoma, 12% were located on the skull and it
was reported at lower jaw (27%), maxillary (22%), spine (15%), ribs (10%), nasal cavity and paranasal sinuses (9%) and pelvis
(5%) (Gomes et al., 2008, Rodrigues et al., 2009). The cranial osteosarcoma features has a high rate of
metastasis (Neuwald et al., 2006). Regardless of the therapeutic protocol chosen, the lifetime is estimated at
six months to one year (Cavalcanti et al., 2004). The metastases signals tend to appear quickly causing
lethargy, anorexia, weight loss (Gomes et al., 2008). In most cases, euthanasia is chosen for these
patients (Daleck et al., 2006, Gomes et al., 2008, Neuwald et al., 2006). Diagnosis is based on clinical history,
physical examination, radiological and scintigraphic findings, and the confirmation made by biopsy and
histopathology. The cytology by fine needle aspiration can be a form ofdiagnosis in some cases
being a less invasive and lowest cost method
(Oliveira and Silveira, 2008). The differential diagnosis includes
other bone neoplasia, osteomyelitis, bone repair, bone diseases and
metabolites periostitis, hypertrophic pulmonary osteoarthropathy, and
panosteitis metafisial osteopathy (Souza et al., 2008). The clinical signs of axial
osteosarcoma depend on the anatomic development and volume and may cause dysphagia, exophthalmos, pain on opening the
mouth, facial deformity, nasal discharge, sneezing, soreness and signs of neurological disorder (Gomes et al., 2008).
Surgical treatment is not always effective since depending of their location it may becomes difficult to obtain edges free of neoplastic cells, and may be
associated with chemotherapy and / or radiation therapy (Neuwald et al., 2006). The combination of surgery with chemotherapy has been
the treatment protocol that provides improved survival (Oliveira and Silveira, 2008).
The drug indicated for chemotherapy is the carboplatin (Rodrigues et al., 2009). Pain control
is very important for a patient having a neoplasm; it can be administered as an analgesic or nonsteroidal anti-inflammatory
opioids (Daleck et al., 2009). The objective of this study is to report a osteosarcoma in the axial skeleton case, at the cranial region, in a large sized dog, male, who showed no
metastasis confronting some literature data.
Case report
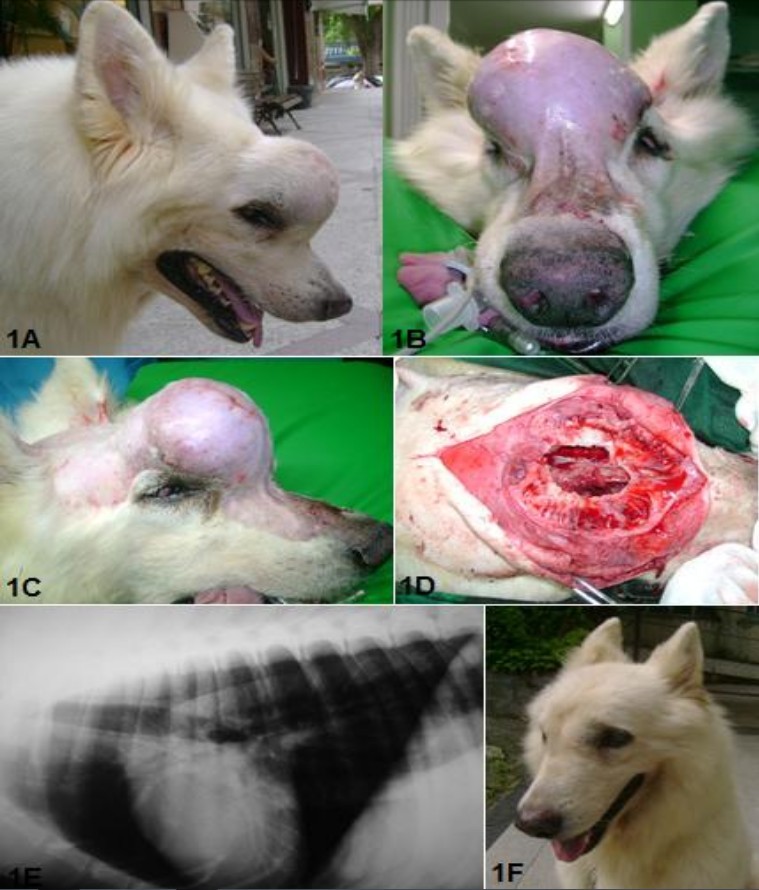
A male White Shepherd, 8 years old was referred for evaluation,
diagnosis and surgical intervention, with a slight elevation in the frontal region of the skull, an
increase of 3 weeks (Figure 1A, 1B and 1C). Upon examination, it was observed that the patient, though having slight deformity of
the face showed no clinical signs of involvement of the region of the paranasal sinuses such as sneezing and nasal discharge. It was performed a fine needle
aspiration cytology (FNAC), which was inconclusive.
The patient underwent ultrasound
examination and radiographs of the chest and skull, which showed discrete osteolytic destruction in the frontal bone of the skull,
pulmonary imaging with no markings and the presence of anechoic areas in the spleen. A surgical procedure
was performed to remove material for histopathological examination of the frontal region and
for splenectomy and exploratory laparotomy for evaluation of the abdominal
cavity. The histopathology confirmed the presence of osteosarcoma in the frontal and nasal bones. The
patient then underwent surgical resection of the tumor with ostectomy for border security in the
region and curettage of frontal sinus and choanal who presented with purulent sinusitis (Figure 1D). A drain tube was
placed in the nasal cavity for irrigation daily (maintained for 5 days) to avoid the formation of clots and occlusion of the
nasal conduit. Anti-inflammatory and analgesic drugs were administered for five days after surgery.
The stitches were removed 15 days postoperatively, and the pacient was evaluated by clinical, laboratory and imaging examination, prior
to the start of chemotherapywith carboplatin. The patient was followed with monthly clinical and radiographic
evaluation (Figure 1E and 1F). After four months of surgery, there was a recurrence of the tumor that affected
the orbit and adjacent region. The evaluation and staging revealed
no signs of metastasis. The dog was maintained on analgesic and antiinflammatory
treatment; associating Dipyrone (25mg/kg/vo) every 08 hours, alternating with Tramadol (4mg/kg/vo) or morphine
(1.0 mg
/kg/po) every 08 hours and meloxicam (0.1mg/kg/vo) every 24
hours. The patient was euthanized at 10 months after surgery.
Results an Discussion
Osteosarcoma is the most common primary bone cancer diagnosed in dogs (Neuwald et al., 2006), however,
the incidence in the axial skeleton is 25% (Gomes et al., 2008, Oliveira and Silveira, 2008,Cavalcanti et
al., 2004), being the female twice affected comparedwith the occurrence in males (Daleck et al.,
2006), and in older dogs and midsize. The patient reported was a large dog and going into the senile
age.
The location of the lesion in the frontal region, involving the paranasal sinuses and nasal cavity, corroborates with the literature (Gomes et al., 2008,
Rodrigues et al., 2009) reported that the incidence of 9% of cases with this region.
Initial clinical signs with increased frontal region leading to facial
deformityradiographic evidence of bone osteolysis and frontal sinusitis indicated osteosarcoma (Oliveira and Silveira,
2008). The sinusitis is secondary to tumor obstruction in the
nasal cavity that prevents drainage of the sinuses to the middle nasal meatus. However, except for the increase in
volume, this patient had no dysphagia, exophthalmos, pain on opening the mouth, nor signs
of neurological disorder (Gomes et al., 2008) as reported in the literature. A
fine needle aspiration cytology although being a less invasive and less expensive exam
(Oliveira and Silveira, 2008), was inconclusive for this
case, and the patient was referred for biopsy. The diagnosis was confirmed by the gold standard test
for cancer, which was histopathology (Gomes et al., 2008,Oliveira and Silveira, 2008).
Although the effectiveness of surgical treatment is limited by the location of the tumor, the
combination with chemotherapy and/or radiation (Neuwald et al., 2006) may improve the prognosis generating
longer survival times (Oliveira and Silveira, 2008). This dog, 15 days after surgery, underwent
a protocol of carboplatin (Rodrigues et al., 2009) every 21 days. Since he showed
hematological abnormalities, the treatment was interrupted after the
second session. This interruption in the chemotherapy protocol may have contributed to tumor recurrence after 4
months of surgery, and the patient was euthanized at 10
months postoperatively (Daleck et al., 2006, Gomes et al., 2008, Neuwald et al., 2006,5). The euthanasia was an owner's option because
of the tumor extension into adjacent orbit regions, making the surgical procedure not viable.
Pain control (Rodrigues et al., 2009) was satisfactory in this patient with the
combination of ananalgesic (dipyrone), an opioid (tramadol / morphine) and an anti-inflammatory
(Meloxicam). This outcome was measured by daily observation of feed, activities, interaction with the owner, and physiological signals.
Until the euthanasia, the patient was orexigenic with normodipsia and normuria.
However, usually patients with axial skeleton tumor classified as cranial osteosarcoma have a high rate of metastasis
(Neuwald et al., 2006), a fact that was not observed during follow-up of this patient, even with the same tumor
recurrence, the patient continued to feed and daily activities with no clinical signs of lethargy, anorexia, weight loss (Gomes et al.,
2008). The patient was monthly evaluated with chest radiography and abdominal ultrasonography, and was not observed evolution of
possible outbreaks of micrometastases, which are
reported as common in patients with osteosarcoma.
Conclusion
The axial skeleton osteosarcoma is a locally invasive neoplasm and very little metastatic, as reported in
this case. The prognosis is guarded to grave, with frequent euthanasia in patients with recurrences due
to the extension of the neoplastic lesion.
Share this article / Teilen Sie diesen Artikel
Diese Webseite wurde mit Jimdo erstellt! Jetzt kostenlos registrieren auf https://de.jimdo.com








