
Article added / Artikel hinzugefügt 01.10.2021
Generally Articles and Discussions about Osteosarcoma in Dogs
→ Evaluations of phylogenetic proximity in a group of 67 dogs with
osteosarcoma: a pilot study
Article added / Artikel hinzugefügt 01.10.2021
Generally Articles and Discussions about Osteosarcoma in Dogs
→ Canine Periosteal Osteosarcoma
Images added / Abbildungen hinzugefügt 02.05.2019
Generally Sonography Atlas of Dogs →
Cardiovascular system → Pulmonary vessels
New subcategory added / Neue Unterkategorie hinzugefügt 02.05.2019
Generally Sonography Atlas of Dogs →
Cardiovascular system → Pulmonary vessels
Images added / Abbildungen hinzugefügt 01.05.2019
Generally Sonography Atlas of Dogs →
Cardiovascular system → Heart valvular diseases



Targeted radiotherapy with Sm-153-EDTMP in nine cases of canine primary bone tumours

MILNER, R., DORMEHL, I., LOUW, W., CROFT, S.. "Targeted radiotherapy with Sm-153-EDTMP in nine cases of canine primary bone tumours".

ABSTRACT
Nine dogs with primary bone tumours were treated with Samarium-153-EDTMP (Sm-153-
EDTMP). Conventional treatment protocols were precluded by the size of the dogs and the
owners’ refusal of limb amputation. All the tumours were of the appendicular skeleton;
4 were confirmed osteosarcomas. The other 5 tumours were radiologically suspect for
osteosarcoma. Bone scans were performed on all dogs using Technetium-99m-methylene
diphosphonate (Tc-99m-MDP) before administration of Sm-153-EDTMP. Regions of
interest were identified over the contralateral limb at the same site as the tumour and counts
per pixel were recorded for the tumour and contralateral limb and expressed as a ratio. The
dogs were given 1 injection of 37 MBq/kg (1 mCi/kg) of Sm-153-EDTMP intravenously.
Thoracic and primary tumour site radiographs were taken at monthly or 2-monthly
intervals to monitor progression of the primary tumour and search for evidence of
metastasis. Two dogs showed no response to treatment, with an increase in bone pain, and
were euthanased within 1 month. In 1 dog, a tumour of the scapula underwent complete
involution and the dog is considered free of disease at 20 months post Sm-153-EDTMP
treatment. The overall tumourcidal effect of a single dose of Sm-153-EDTMP on primary
bone tumours was difficult to evaluate in this group of dogs, as, with one exception, all the
primary tumours progressed over time and the dogs were euthanased. Pain control, for
which Sm-155-EDTMP is used in man, was not evident, except in the dog that responded
completely to treatment.
Key words: canine, osteosarcoma, radiotherapy, Sm-153-EDTMP.
INTRODUCTION
Neoplasia of the skeleton occurs in numerous vertebrate species, although the dog has a higher incidence
(6.5 in 100 000) than other domestic species and man20,23. Primary skeletal neoplasia accounts for 80 % of these cases. Biphasic incidence of
osteosarcoma occurs in large breeds of dogs, with a peak at 18–24
months and, in older dogs, a median of 7 years 11,20,25. Osteosarcoma in the dog has a predilection for the appendicular skeleton with
the majority of tumours occurring in the metaphyseal area (75 %)11,25
Treatment of osteosarcoma in the dog involves a multi-modality approach. Amputation of the effected limb has been reported to give a mean survival time of 19.8 weeks with 11.5 % alive at 1 year
and 2 % at 2 years21. Currently the ‘gold standard’ appears to be amputation combined with cisplatin chemotherapy, resulting in a median survival rate of 46.4 weeks and a 1 or 2 year survival
rate of 45.5 % and 20.9 % respectively2. A recent article has reported similar survival rates using doxorubicin and surgery3. There are, however, a large number of dogs in which amputation is
precluded by various factors, for example the size of animal. Heidner et al.9 explored the use of cobalt 60 radiation and intra-arterial cisplatin without amputation. They reported a median
survival time of 34.3 weeks, which rose to 46.9 weeks when dogs with metastases were excluded.
Unfortunately this treatment option is not always available. Other treatment options include beta-emitting radio-isotopes coupled to bone-localising pharmaceuticals. To be effective, these
isotopes and pharmaceuticals must fulfil certain requirements: minimum chemical toxicity; half-life of medium duration (approximately 2–8 days) so as to provide optimum radio-biological effect;
the radio pharmaceutical must have a high affinity for diseased bone in relation to normal bone; minimal deposition outside the skeleton and within bone marrow; and the mode of radioactive decay
should be principally beta-particle emission so that the deposition of energy is concentrated
in the immediate vicinity of a lesion13. Samarium-153-ethylenediaminetetramethylene phosphonic acid (Sm-153-EDTMP) fulfils these criteria. Samarium-153 is a medium-energy beta-emitting
radio-isotope (Ebmax = 0.80 MeV, T =
46.8 h) with a gamma photon (abundance 28 %, KeV 103) coupled to the diphosphonate EDTMP13,14. Lattimer et. al.12 reported the use of Sm-153-EDTMP in canine bone tumours. Forty dogs were treated
with a single intravenous dose of 37 MBq/kg (1 mCi/kg); 20 dogs received an additional intravenous dose 1 week later. The single dose was calculated to expose the neoplasm to approximately 40 Gy
of radiation. The results were variable. Seven dogs were regarded as disease-freed, 25 responded partially, and 8 did not respond. Those with a complete or prolonged response belonged to the
single-dose group. The best response was noted in small lesions with minimal lysis, metastatic lesions and axial skeletal neoplasia. It is worth noting that Sm-153-EDTMP was not the only
treatment
administered in these cases.
Moe et al.19 described treatment of an osteosarcoma of the maxilla with Sm-153-EDTMP combined with surgical debulking of the mass. No local recurrence or metastases were evident 21 months postsurgery. Straw et al.22 reported the use of Sm-153-EDTMP in 2 dogs with mandibular osteosarcoma. One dog was lost to follow-up at 41 months. The other dog died of renal failure at 6.9 months, with recurrence of the tumour at the primary local site.
In humans, Sm-153-EDTMP is used extensively for palliation of bone pain
caused by metastatic bone cancer1. Bruland et al.4 recently reported good transient response to treatment of a
human vertebral primary osteosarcoma with Sm-153-EDTMP.
Complications of Sm-153-EDTMP treatment are myelotoxicity1,12,13 and, in some cases, an increase in bone pain
following treatment (flare response)1,14. Myelotoxicity occurs 1 week after treatment and remains haematologically
evident for 2–3 weeks12,13. Renal toxicity does not appear to be significant, although Sm-153-EDTMP, like other
diphosphonates, is cleared by the kidneys7. Acute radiation nephritis is considered to occur only after exposure
to radiation of more than 10 Gy17. Lattimer et al.12 did not report renal toxicity.
On the basis of the above reports, we concluded that Sm-153-EDTMP might be of some benefit (pain control, tumourcidal effect) in dogs that could not undergo conventional therapies for bone tumours. The results of targeted radiotherapy in 9 dogs are reported.
MATERIALS AND METHODS
All dogs considered to be possible candidates underwent full clinical examinations. Radiographs were taken of
the primary tumour site, as well as the following thoracic radiographs: left and right lateral, dorsoventral and ventrodorsal views. Only dogs free of radiological evidence of lung metastases and
with signed consent of their owners were selected for treatment (n = 9). The mean age of the dogs was 5.56 years (SD ± 2.44) and mean weight 41.4 kg (SD ± 9.88). There was a bias towards heavier
animals, as these dogs could not undergo amputation. The size of primary tumours were recorded from radiographs. The measurements were made sequentially as follows:
length of the tumour along the long axis of the bone, maximum measurement in a cranio-caudal orientation, lateral to
medial (maximum) measurement (always at 90° to the former measurement). The margins were defined as those areas showing abnormal bone changes associated with bone tumours. The cases were then
staged using the TNM method for bone tumours10, where T0 = no evidence of a primary tumour, T1 = tumour confined to within bone medulla/cortex, T2 = tumour extending beyond periosteum, M0 = no
evidence of metastases and M1 = distant metastases present.
In all the dogs, the affected limb was lame and non-weight-bearing. Four dogs (see Table 1; Cases 3, 4, 6, 7) had
osteosarcomas confirmed by biopsy or at necropsy. The other 5 dogs had tumours considered to be radiographically typical for appendicular osteosarcoma (site, radiographical appearance,
progression with metastases obvious at euthanasia) except case 9, which had a neoplasm of the scapula that was classified as an aggressive primary bone tumour involving the entire wing of the
scapula. All dogs underwent an initial bone scan using technetium-99m-methylene diphosphonate (Tc-99m-MDP) (Atomic Energy Corporation of South Africa) as an indicator of tumour uptake by
Sm-153-EDTMP to predict the likelihood of successful treatment12. The intravenous dose of Tc-99m-MDP varied between 185 and 592 MBq depending on body weight. Three hours post-injection, light
sedation
was achieved using metomidine HCl (Domitor 1 mg/ml, Ciba) at 0.1 ml/10 kg and thiopentone sodium. Scanning was
then immediately performed using a Siemens Orbital gamma camera (low energy collimator) (energy peak 140 keV,
window 15 %). Two-minute acquisition times were used and the data captured in 64 × 64 word mode. For comparative purposes, a reference region of interest (ROI) was identified at the tumour site
and at the same location on the contralateral limb. Counts per pixel were recorded for the tumour (T) and contralateral ROI (NTC = non-tumour counts) and calculated as a ratio (T/NTC) in Table 1.
Results of Case 3’s Tc-99m-MDP scan were not recorded, as these were lost.
Sm-153-EDTMP (Atomic Energy Corporation of South Africa) was administered to all 9 dogs 7 days later (for logistical
reasons) at a dosage rate of 37 MBq/kg intravenously over a 30-second period via an indwelling catheter. The catheter was flushed after administration of Sm-153-EDTMP with 5 ml sterile saline
solution to ensure complete administration of the calculated dose. All cases returned monthly or bimonthly for follow-up radiographs of the thorax and primary
tumour site. The size of the primary tumour and evidence of metastases were recorded. Blood samples were collected from the cephalic vein in EDTA tubes (Becton Dickinson, Vacutainer Systems,
Europe) for haematology after treatment with Sm-153-EDTMP in 2 dogs (Cases 6, 8) at 2 and 4 weeks post-treatment. Blood samples of the other 7 dogs were not taken for various reasons such as
owner non-compliance, distance from the academic hospital, and cost of tests.
For ethical reasons all the dogs were dosed with 0.3 mg/kg of a non-steroidal anti-inflammatory substance, piroxicam
(Feldene, Pfizer Laboratories), once every 2nd day, for pain control, and 2–5 µg/kg of misoprostil (Cytotec, G D Searle) twice a day to prevent gastric ulceration. These medications were
administered for at least 14 days before radio-isotope treatment, to distinguish between pain control by piroxicam and by Sm-153-EDTMP. The owners were requested to monitor improvement in limb
function over time, and to note other signs associated with pain, such as loss of appetite.
RESULTS
The results of the Tc-99m-MDP bone scan and other data are presented in Table 1. Clinical staging in all dogs except
Case 8 identified single primary tumours not confined to the medulla and cortex with no evidence of metastasis, which were thus staged as T2M0. Case 8 was staged as T1M0, as the tumour was still
confined to the medulla and cortex. All cases except 1, 2 and 9, which were euthanased within 1 month (1, 2), or underwent involution of the tumour (9), developed progressive enlargement of the
primary tumour with time. However, in the tumours in Cases 7 and 8 more sclerotic bone was evident radiographically, and these tumours seemed to progress more slowly. Case 7, a female Rottweiler,
underwent amputation (no chemotherapy) of the affected limb 4 months after treatment, owing to progressive enlargement of the tumour. At the time when surgery was performed no evidence of
metastasis was present. Two months later she was admitted with widely disseminated metastases in most organs, including muscle tissue, and was euthanased. All other cases except Case 9 were
eventually euthanased. Necropsies were performed on 3 dogs (Cases 4, 6, 7). Only Case 6 had no evidence of macro- or microscopic metastases.
Pain control was difficult to evaluate, since all dogs had advanced tumours of weight-bearing limbs and were lame.
Case 9, which is considered to be diseasefree at this stage12, was the only exception. This dog responded rapidly to treatment, with good control of pain, within a 2-week period post-treatment.
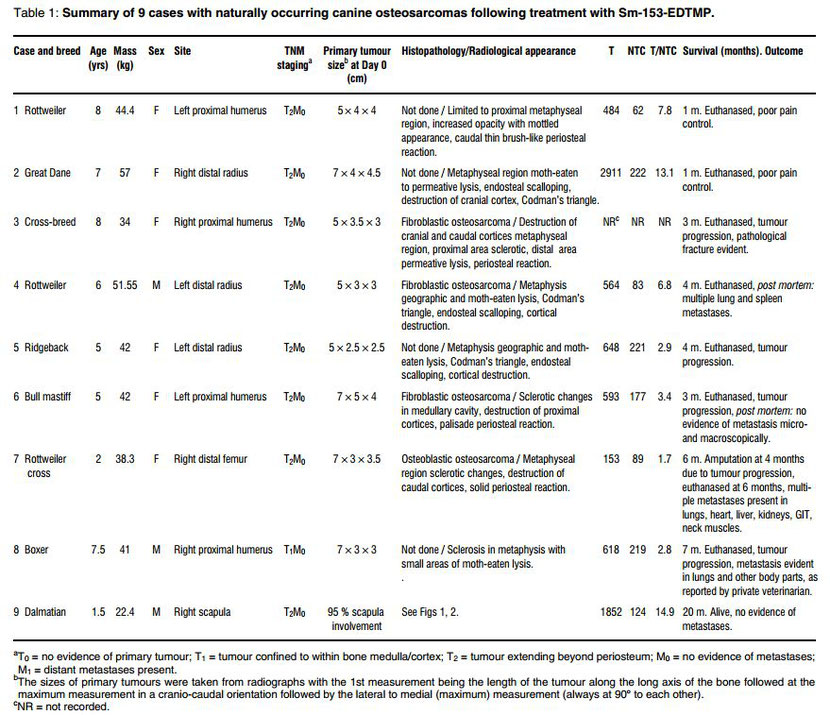
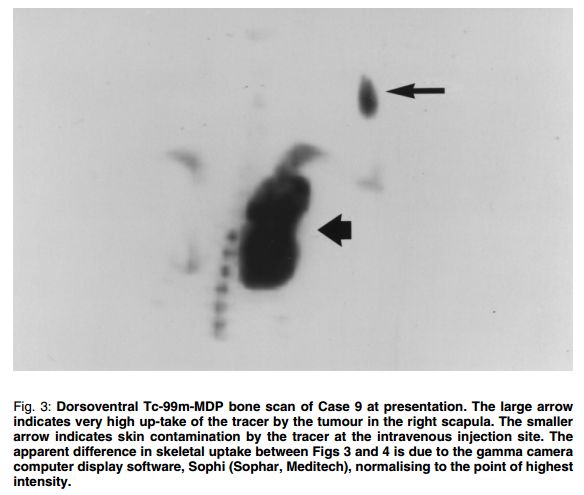
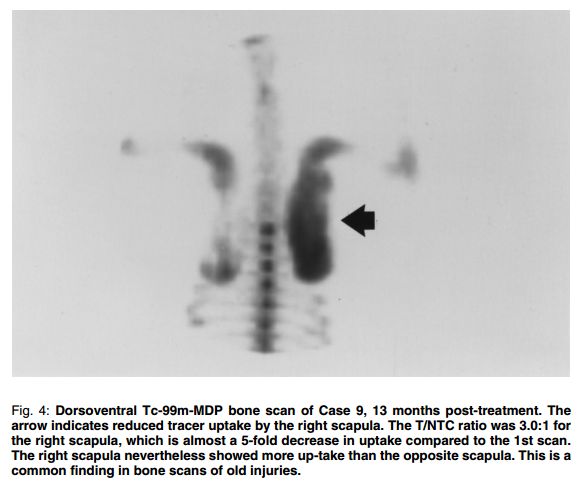
Figures 1 and 2 are radiographs of the scapular tumour (Case 9) on initial presentation and 13 months post-treatment respectively. Figure 3 is a dorsoventral view of the Tc-99m-MDP study illustrating the extensive involvement of the right scapula. Figure 4 is a follow-up Tc-99m-MDP bone scan 13 months posttreatment.
Haematological findings in Cases 6 and 8 included leukopaenia and thrombocytopaenia at 2 weeks, with a return to
normal within 4 weeks. These findings are in agreement with reports in the literature on the effects of Sm-153-EDTMP on blood parameters.
DISCUSSION
All the dogs except Case 8 (Table 1) had tumours extending beyond the periosteum, which according to the report by
Lattimer et al.12 should be associated with a poor response to Sm-153-EDTMP treatment. Our findings, with the exception of 1 dog (Case 9), support this, as all these tumours progressed, resulting
in euthanasia of the dog. After the Sm-153-EDTMP injection, the tumour of the scapula (Case 9) underwent rapid involution that was radiologically evident within 3 months. The right forelimb is
fully functional and no metastatic lesions are evident to date (20 months post-treatment) in thoracic radiographs or whole body bone scan. Lattimer et al.12 reported one case that also responded
rapidly, with complete involution of the tumour. The tumour was reported to be a chondrosarcoma of the humerus. In a recent report of osteosarcomas of the scapula, high prevalence of metastasis
was found, with
a short overall survival time (17 weeks)8. Partial scapulectomy combined with cisplatin therapy of primary bone neoplasia involving the proximal half of the scapula has also been reported; most
cases developed metastatic lesions24. Case 9 is therefore unique, as the tumour involved the whole scapula (Fig. 1), and to date there is no evidence of primary tumour recurrence or metastases.
A relationship between radiographical appearance and uptake of Sm-153-EDTMP as reflected by the T/NTC ratio
seems to suggest a higher ratio in tumours with more lytic (radiographic) lesions (Cases 1, 2, 3, 4, 5) than those with sclerotic changes (6, 7, 8). This might be explained by the fact that bone
lysis is more indicative of an aggressive lesion that may have more reactive tissue and a greater blood supply and therefore accumulate more isotope6,18. However, Lewington14 suggested that
therapy will be more effective in predominantly sclerotic lesions with a brisk osteoblastic reaction than in more destructive lytic lesions. It is interesting to note that the 3 dogs (6, 7, 8)
with radiological evidence of sclerosis had lower T/NTC ratios, but survived longer. Forrest et al.6 reported that a high pretreatment count per pixel signifies an aggressive tumour subject to
early metastasis, which may explain the poorer
response in Cases 1, 2 and 3. The exception was the scapular tumour in Case 9, which showed the highest T/NTC ratio, but which underwent complete involution post-treatment.
Some dogs (Cases 1 and 2) experienced an increase in bone pain following injection of Sm-153-EDTMP. This is known in man as a flare response and is seen soon after administration of the radio-pharmaceutical (3 days)14. The response is thought to be due to radiation-induced endosseous oedema and is, ironically, regarded as a good prognostic sign since these patients reported dramatic pain relief subsequently14. Lewington14 reported that pain relief is independent of tumour radiosensitivity, and therefore one should be wary of interpreting control of pain as a reflection of the radio-isotope’s tumourcidal effect.
The exact dose received by each tumour in this study is not known, but Lattimer et al.13 reported that when the lesion to normal bone ratio was 16:1, as determined by acute drill hole model, the dose delivered to the lesion was approximately 40 Gy (skeletal weight of the dog taken as 100 g/kg, where <1 % of the skeleton is involved). This is similar to the total dose used in external beam radiation of appendicular osteosarcoma; however, the dose was delivered in 10 fractions using a Monday, Wednesday and Friday protocol9. Only 2 of our dogs (Cases 2, 9) had T:NTC ratios approximating the reported ratio, 13.1 and 14.9 respectively, one of which (Case 9) is regarded as cured.
It is obvious that where the tumour is susceptible to the radiotherapy, as in Case 9, a single dose of 37 MBq/kg
Sm-153-EDTMP was adequate to achieve remission of the tumour. However, in the other 8 dogs the results were poor.
It also appears that the diphosphonate, EDTMP, has pharmacological activity that may influence radio-pharmaceutical
localisation in neoplastic tissue14,15. Dormehl et al.5 found that repeated doses of Sm-153-EDTMP in the baboon led to progressively decreased uptake in normal bone, and this is thought be
due to the blocking effect of the diphosphonate EDTMP.
CONCLUSION
While in this study the results obtained with Sm-153-EDTMP in spontaneously occurring canine osteosarcomas of the
appendicular skeleton to control bone pain and/or tumour growth were spectacular in 1 dog, the overall results for the other 8 dogs were inconclusive, and further investigation is required.
Evaluation of Sm-153-EDTMP combined with other treatment modalities, as reported by Moe et al.19, may hold more promise, as well as investigation into other beta-emitting
radio-pharmaceuticals14,16.
ACKNOWLEDGEMENTS
We thank the radiology section of the Onderstepoort Veterinary Academic Hospital for taking and processing the
radiographs and dedicate this article to the memory of Maria Jimenez who was part of that department. A special word of thanks is due to the following owners and their special dogs: N J
Liebenberg (Jessica), E E K Jarvis (Zeus), L Lemmer (Umfubu), T L Pretorius (Chica), E Mobbs (Ascombe), H J du Plessis (Max), I van den Ende (Zack), A D Argyle (Cambel) and A W Hancock (Sheba).
REFERENCES
1. Bayouth J E, Macey D J, Kasi L P, Fossella
F V 1994 Dosimetry and toxicity of Samar-
ium-153-EDTMP administered for bone
pain due to skeletal metastases. The Journal
of Nuclear Medicine 35: 63–69
2. Berg J, Weinstein M J, Schelling S H, Rand
W M 1992 Treatment of dogs with osteosar-
coma by administration of cisplatin after
amputation or limb-sparing surgery: 22
cases (1987–1990). Journal of the American
Veterinary Medical Association 12: 2005–2008
3. Berg J, Weinstein M J, Springfield D S, Rand
W M 1995 Results of surgery and doxoru-
bicin chemotherapy in dogs with osteosar-
coma. Journal of the American Veterinary
Medical Association 206: 1555–1560
4. Bruland O S, Skretting A, Aas M 1996
Targeted radiotherapy of osteosarcoma
using 153 Sm-EDTMP. A promising
approach. Acta Oncologica 35: 381–384
5. Dormehl I, Louw W K A, Schneeweiß F,
Milner R J, Schmitt G, Croft S 1997 Uptake
of samarium-153-ethylenediaminetetra-
methylene phosphonic acid in normal bone
after multiple applications: a primate study.
Arzneimittel-Forschung Drug Research (In
press)
6. Forrest L J, Dodge R K, Page R L, McEntee
M C, Novotney C A, Thrall D E 1992
Relationship between quantitative tumour
scintigraphy and time to metastasis in dogs
with osteosarcoma. The Journal of Nuclear
Medicine 33: 1542–1547
7. Goeckeler W F, Edwards B, Volkert W A,
Holmes R A, Simon J, Wilson D 1987 Skeletal
localization of Samarium-153 chelates:
potential therapeutic bone agents. The Jour-
nal of Nuclear Medicine 28: 495–504
8. Hammer A S, Weeren F R, Weisbrode S E,
Padgett S L 1995 Prognostic factors in dogs
with osteosarcoma of the flat or irregular
bones. Journal of the American Animal Hospi-
tal Association 31: 321–326
9. Heidner G L, Page R L, McEntee M C,
Dodge R K, Thrall D E 1991 Treatment of
canine appendicular osteosarcoma using
cobalt 60 radiation and intraarterial
cisplatin. Journal of Veterinary Internal
Medicine 5: 313–316
10. Henderson R A, Brawner W R, Brewer W G,
Henry C J 1995 Clinical staging. In Hahn
K A, Richardson R C (eds) Cancer chemo-
therapy: a veterinary handbook. Williams &
Wilkins, Baltimore: 23–44
11. LaRue S M, Withrow S J 1989 Tumors of the
skeletal system. In Withrow S J, MacEwen E
G (eds) Clinical veterinary oncology. J B
Lippincott Company, Philadelphia: 234–
252
12. Lattimer J C, Corwin L A, Stapleton J,
Volkert W A, Erhardt G J, Ketring A R,
Anderson S K, Simon J, Goeckeler W F 1990
Clinical and clinicopathologic response of
canine bone tumor patients to treatment
with Samarium-153-EDTMP. The Journal of
Nuclear Medicine 31: 1316–1325
13. Lattimer J C, Corwin L A, Stapleton J,
Volkert W A, Erhardt, G J, Ketring A R,
Hewett J E, Goeckeler W F 1990 Clinical and
clinicopathologic effects of Samarium-153-
EDTMP administered intravenously to
normal Beagle dogs. The Journal of Nuclear
Medicine 31: 586–593
14. Lewington V J 1996 Cancer therapy using
bone-seeking isotopes. Physics in Medicine
and Biology 41: 2027–2042
15. Lin J H 1996 Biphosphonates: a review of
their pharmacokinetic properties. Bone 18:
75–85
16. Lin W, Lin C, Yeh S, Hsieh B, Tsai Z, Ting G,
Yen T, Wang S, Knapp F F, Stabin, M G 1997
Rhenium-188 hydroxyethylidene diphos-
phonate: a new generator-produced
radiotherapeutic drug of potential value
for the treatment of bone metastases.
European Journal of Nuclear Medicine 24: 590–
595 (Abstract)
17. Madrazo A, Schwarz G, Churg J 1975
Radiation nephritis: a review. Journal of
Urology 114: 822–827 (Abstract)
18. Mettler F A, Monsein L, Rosenberg R D 1988
Radionuclides and the normal bone scan. In
Mettler F A (ed.) Radionuclide bone imaging
and densitometry. Churchill Livingstone,
New York: 13–30
19. Moe L, Boysen M, Aas M, Lonaas L, Gamlen
H, Bruland O S 1996 Maxillectomy and
targeted radionuclide therapy with
153Sm EDTMP in a recurrent canine
osteosarcoma. Journal of Small Animal
Practice 37: 241–246
20. Pool R R 1990 Tumors of bone and cartilage.
In Moulton J E (ed.) Tumors in domestic
animals. University of California Press,
Berkeley: 157–227
21. Spodnick G J, Berg J, Rand W M, Schelling
S H, Couto G, Harvey H J, Henderson R A,
MacEwen G, Mauldin N, McCaw D L,
Moore A S, Morrison W, Norris A M, O’Bra-
dovich J, O’Keefe D A, Page R, Ruslander D,
Klausner J, Straw R, Thompson J P,
Withrow S J 1992 Prognosis for dogs with
appendicular osteosarcoma treated by am-
putation alone: 162 cases (1978–1988). Jour-
nal of the American Veterinary Medical
Association 200: 995–999
22. Straw R C, Powers B E, Klausner J, Hender-
son R A, Morrison W B, McCaw D L, Harvey
H J, Jacobs R M, Berg R J 1996 Canine
mandibular osteosarcoma: 51 cases (1980–
1992). Journal of the American Animal Hospital
Association 32: 257–262
23. Theilen G H, Madewell B R 1987 Tumors of
the skeleton. In Theilen G H, Madewell B R
(eds) Veterinary cancer medicine. Lea &
Febiger, Philadelphia: 471–497
24. Trout N J, Pavletic M M, Kraus K H 1995
Partial scapulectomy for management of
sarcomas in three dogs and two cats. Journal
of the American Veterinary Medical Association
207: 585–587
25. Waters D J 1993 Musculoskeletal system. In
Slatter D (ed.) Textbook of small animal
surgery. W B Saunders Company, Philadel-
phia: 2213–2230
Share this article / Teilen Sie diesen Artikel
Diese Webseite wurde mit Jimdo erstellt! Jetzt kostenlos registrieren auf https://de.jimdo.com








